 /caesium-cs--chemical-element--3d-rendering-isolated-on-black-background-922704282-5adb6de08e1b6e0037170151.jpg)
atoms can combine in ratios to form compounds.all atoms of an individual element have the same properties including mass.that elements were made of small indivisible particles called atoms.Mendeleyev predicted the existence of previously unknown elements and paved the way for modern chemistry. The first periodic table that ordered the elements based on their atomic weight was developed by Dmitri Mendeleyev, a Russian chemist, in the 1860s. The last lessons concentrated on the chemical and physical aspects of biology. We have covered an extensive (but not exhaustive) review of the main disciplines of the sciences: scientific investigation, physics, chemistry, Earth science, and biology. The material will concentrate on empirical characteristics of the elements that are reflected by their valance electrons. In this lesson, we will be reviewing the properties of the periodic table and the patterns of the elements displayed therein. 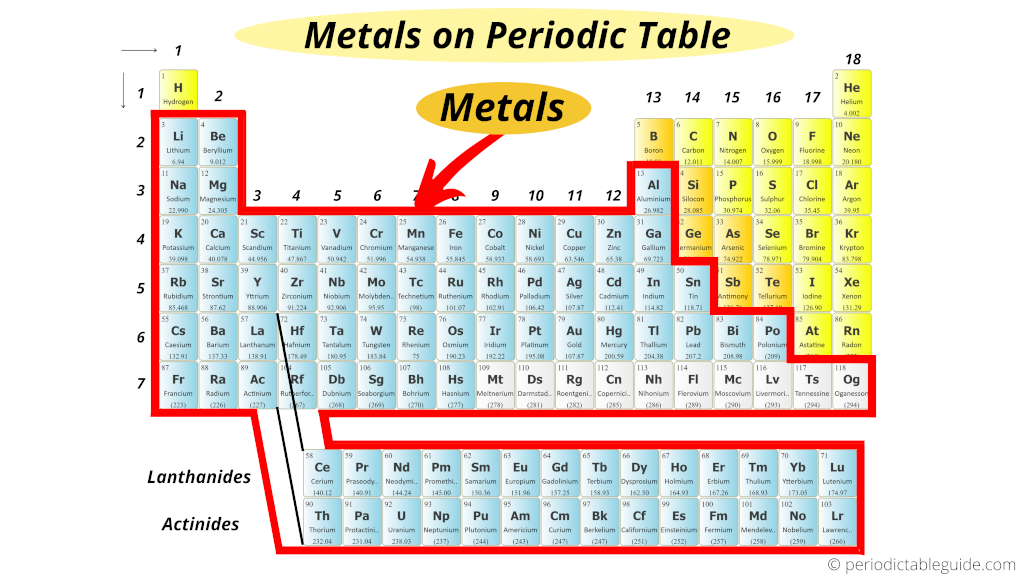
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
